|
12/15/2023 0 Comments Carbon electron configuration drawing  
So Lithium's nextĮlectron, as you build up the Lithium atom must go In terms of energy, so that would be the two s orbitals. It's third electron has to go into this next highest orbital We've taken care of the two of it's three electrons. Doesn't really matter which one is which. So one of them is, it would be two px, one of them would be two py and one of them would be two pz. They're of higher energy so here are the two p orbitals and So we draw in our p orbitals in the second energy level. Then we also have p orbitals in the second energy level, we have three of them. Next let's draw in this s orbital in the second shell, So we already drew in this s orbital in the first shell. So we have three p orbitals in the second energy level as well. So three possible values means three p orbitals here. The allowed values for ml would be negative one, zero and positive one. So in the second shell, in the second energy level, we also have an s orbital and we also have one of them and we also have to think about l is equal to one, that's So when l is equal to zero, we're talking about an s orbital. So we talked about that again in the videos on quantum numbers. What are the allowed values for l? L could be equal to zero or l could be equal to one. So in the second shell, n is equal to two. So we have to move on to the second shell to add in Lithium's third electron. 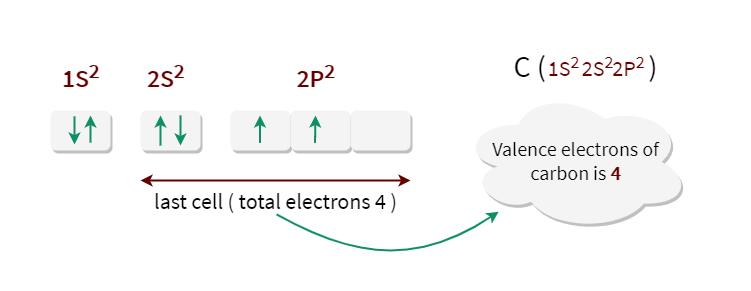
Well Lithium has three electrons but the first shell is full, it's closed. So writing the electronĬonfiguration for Lithium, let me go ahead and we'll start writing it right here. The first two electrons for Lithium are going to go into this one s orbital. And there's only one s orbital in the first shell here so I can draw in, let me go ahead and draw that orbital in. The first shell when n is equal to one, the only possible value for l is zero. So we talked about this in the last video. The first two electrons for Lithium are going to go into the first shell. Let's think about the first two electrons for Lithium. So we find the second period on the periodic table and we go across and the first element we see is Lithium with three electrons. Let's do electron configurations for the second period. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
